Particulate Matter Testing
Cindy Pickens, ARL Bio Pharma Laboratory Supervisor
Particulate Matter testing is performed on all parenteral solutions to determine the cleanliness and stability of the solution; and, is listed in USP <797> as a necessary component for quality assurance of compounded sterile preparations under the responsibilities of compounding personnel. It is also specifically outlined as a required test in several individual monographs such as Morphine Sulfate Compounded Injection.
USP Chapter <788> specifically deals with particulate matter in parenteral solutions. The analysis can be carried out in either two methods and are further broken down into categories, ‘a’ and ‘b’, based on the volume of the sample. Method 1 is an instrumental method that utilizes light obscuration on a prescribed volume of solution and is the preferred method of analysis. Method 2 is a manual microscopic method where the sample is manually filtered through a membrane and physically counted utilizing a microscope. Method 2 is used only when Method 1 cannot be performed. Examples of samples that must be analyzed by Method 2 are those that are oil, highly surfactant, or darkly colored. Each method is broken down into category ‘a’ and ‘b’. Samples in containers >100mL are analyzed by category ‘a’ which provides specifications as “particles/mL”. Samples in containers ≤100mL are analyzed by category ‘b’ which provides specification as “particles/container.” Specifications are:
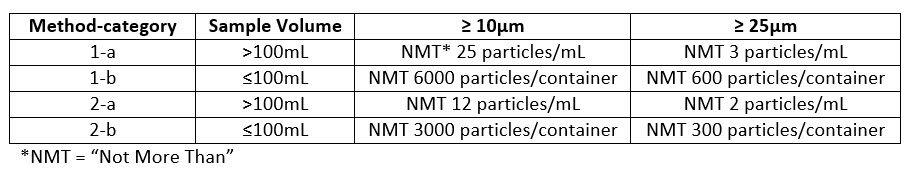
As seen from the specifications, the volume of the sample dictates the format of the results (‘per mL’ or ‘per container’). USP dictates that a minimum of 25mL of sample is required to run a particulate matter test. As long as the sample container has a minimum of 25mL, the sample can be run without further consideration. However, if the container volume is less than 25mL, USP requires that 10 containers must be combined for analysis. If the combined volume of 10 containers is less than 25mL, the sample can be diluted with particle free water so the analysis can be conducted. A dilution factor is applied to allow for an accurate particles/container count.
USP Chapter <789> deals specifically with ophthalmic solutions. In principle, the test is performed in the same manner as <788> Method 1; however there is only one set of limits for ophthalmic solutions regardless of the volume of the sample unless the monograph for that solution dictates otherwise. Additionally, <789> calls for additional testing via Method 2 if it first fails by Method 1.
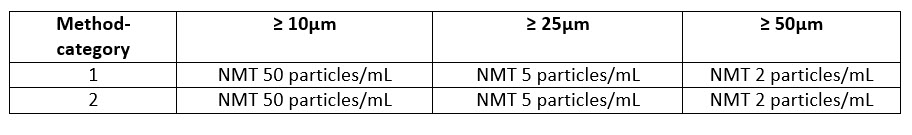
USP Chapter <787> can be used as an alternative for <788> for therapeutic protein injections. This method allows for a lower volume of material to be utilized for testing. The test is performed by Light Obscuration (Method 1) with special consideration given for the sample type, additional system suitability verification steps and blank readings. While some of these samples can be run by method 2, the results are not considered interchangeable as with <788> and <789> due to visual interference of the protein particles and their physical characteristics. The specifications for <787> are equivalent to <788>.

It is important for pharmacists to conduct particulate matter testing on parenteral solutions, as particulate matter in large numbers can cause harm to patients. Testing per USP methods assures compounding personnel that their products meet the requirements for sub-visible particles.
Please contact ARL for more information 800-393-1595 or info@arlok.com.
